Difference Between Ideal Gas And Real Gas
 Rachel Young
Rachel Young 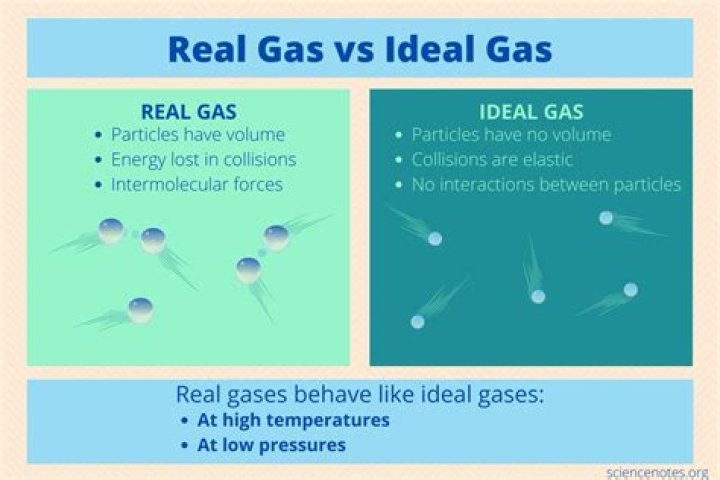
Ideal gases are gases that behave in perfect accordance with the theoretical ideal gas laws, while real gases have slight discrepancies due to various physical effects. These laws deal with the behavior of gases under different physical conditions, and are the foundation for understanding the behavior of the gas. Here we will discuss the main differences between ideal and real gases, and how understanding these differences is important for engineers and scientists.
1. Intermolecular Forces
One of the main differences between ideal gases and real gases is their response to intermolecular forces. Ideal gases do not recognize intermolecular forces at all, while real gases do. This means that the behavior of the real gas is more complicated due to the fact that the molecules of the gas can interact with each other. This has an effect on the physical properties of the gas, such as its compressibility and thermal conductivity.
In a real gas, the molecules of the gas can come into contact with each other and be attracted due to intermolecular forces. This can lead to a decrease in the volume of the gas, as the molecules come into contact and transfer energy. This attraction is not present in ideal gases, and as such the properties of an ideal gas are much simpler than those of a real gas.
2. Compressibility
In ideal gases the molecules are too far apart to experience any attraction due to intermolecular forces, and as a result the gas cannot be compressed. This is not the case for real gases, which have some compressibility due to the intermolecular forces present. Compressibility has a significant effect on the physical properties of a gas, such as its density and enthalpy.
In a real gas the molecules attract each other, and this reduces the volume of the gas while increasing its pressure. This is due to the fact that the molecules are no longer perfectly spaced apart, and as such the pressure increases as they come into contact with each other. Real gases can therefore be compressed, while ideal gases cannot. This makes them more difficult to work with, as they must be compressed in order to use them.
3. Response to Temperature Changes
The response of a gas to temperature changes is also different for ideal and real gases. Ideal gases have no response to temperature changes; they remain the same regardless of the surrounding temperature. Real gases, on the other hand, exhibit a measurable response. As the temperature increases, the molecules of the gas become more energetic, and move around more quickly. This leads to a decrease in pressure, as the molecules are moving faster and are spaced further apart.
The response of a real gas to temperature changes can be used to measure its heat capacity. This is useful in applications such as refrigeration, where the temperature of a gas is changed in order to change its pressure. Knowing the heat capacity of the gas can be important for designing systems with efficient heat transfer.
4. Critical Point
The critical point of a gas is the point at which the liquid and gaseous phases of the gas are indistinguishable from one another. The critical point of an ideal gas is the same as its boiling point, as the gas does not undergo any significant change. Real gases, however, have a different critical point. This is due to the fact that the intermolecular forces present in the gas can cause the molecules to remain attached to each other even after the gas has passed its boiling point.
The critical point of a real gas is therefore different from its boiling point due to the effect of the intermolecular forces. This can be an important factor when working with real gases, as the critical point must be taken into account when determining the temperature at which the gas will begin to condense.
5. Heat Capacity
The affect of temperature on the physical properties of a gas is an important factor, and this is where the heat capacity of a gas comes into play. Ideal gases have a constant heat capacity, as they do not respond to temperature changes. Real gases, however, have an adjustable heat capacity due to their response to temperature changes.
The heat capacity of a gas is an important factor for applications such as refrigeration, where the temperature of the gas must be changed in order to change its pressure. Knowing the heat capacity of the gas is important for designing the system, as it allows the engineer to determine what temperature the gas must be at in order to achieve the desired pressure.
6. Diffusion
Diffusion is the process by which molecules of a gas move from one region of space to another due to their random motion. Ideal gases have constant diffusion rates no matter what the temperature, as they do not experience any changes due to intermolecular forces. Real gases, however, have an adjustable diffusion rate depending on the temperature.
The diffusion rate of a real gas is related to its heat capacity, as the temperature of the gas affects how rapidly the molecules move. This can be important for applications such as chromatography, where the diffusion rate of the gas must be taken into account in order to separate different molecules.
7. Viscosity
The viscosity of a gas is a measure of its resistance to flow. Ideal gases have no viscosity, as their molecules do not experience any intermolecular forces. Real gases, however, have some viscosity due to the intermolecular forces present. This means that their molecules attempt to remain in contact with each other and transfer energy, leading to a resistance to flow.
Viscosity is an important factor for applications such as lubrication, as the viscosity of the gas affects how easily it flows. Knowing the viscosity of a real gas is therefore important when designing a system that uses the gas.
8. Adiabatic Processes
The adiabatic process is the process by which a gas exchanges energy without a change in temperature. Ideal gases undergo the perfect adiabatic process, where they exchange the same amount of energy regardless of their initial state. Real gases, however, have imperfect adiabatic processes, due to the fact that the intermolecular forces cause them to transfer energy at different rates.
The adiabatic process of a real gas is important for applications such as engines, where the efficiency of the system depends on the rate of energy transfer. Knowing the adiabatic process of a real gas is important for designing an efficient system.
9. Experimental Measurements
Experimental measurements are important for characterizing the behaviour of a gas. For ideal gases, these measurements are quite easy to make and interpret, as the equations governing their behaviour are known. For real gases, however, the measurements must take into account the fact that the gas will experience slight deviations due to the intermolecular forces present.
This means that to accurately characterize a real gas, the measurements must take into account the effects of the intermolecular forces. This can be complicated and time consuming, but is necessary in order to gain accurate data about the physical properties of the gas.
10. Summary
Ideal and real gases differ in many ways, due to the effects of intermolecular forces present in real gases. These forces have an effect on the physical properties of the gas, such as its compressibility, viscosity, and diffusion rate. Understanding these differences is important for engineers and scientists, as they must take into account the effect of the intermolecular forces when designing systems that use real gases.



